Using
Saccharomyces cerevisiae to study
cell cycle genes in meiosis
Anne
Galbraith, Dept of Biology, University of Wisconsin-La Crosse.
The growth and division of cells
(mitosis) and the formation of sperm and eggs (meiosis) are important for
almost all organisms, including humans. We all began as a single cell that
resulted from our father’s sperm fertilizing our mother’s egg. This single cell
then divided by mitosis into two cells (daughter cells) which then grew and
divided into four cells (Figure 1A). This process of mitosis continued until we
were born. It then continued again until we were “grown up”. It is still
happening right now as some of our cells use mitosis to replace old dead cells,
such as skin cells. Meiosis, on the other hand, is a special form of mitosis
that occurs only in a special subset of our cells to form eggs and sperm. In
meiosis, one cell divides twice in a row to form four daughter cells from one
cell (Figure 1B). Those cells are then modified to become eggs or sperm.
Mitosis and meiosis, then, are similar processes, but result in very different
types of cells.
A
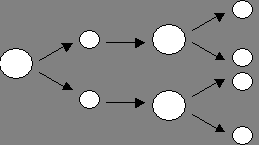
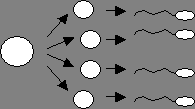
B
Figure
1. A) In mitosis, a single cell (circle on the left) divides to form two
daughter cells. These cells grow, and then divide to form a total of four
cells. Those four cells grow and divide to form eight cells, etc. B) In
meiosis, a single cell divides twice, resulting in four daughter cells that do
not grow and divide again. Instead, these cells are modified to become eggs or
sperm in humans.
One of the most important
requirements of a successful mitosis or meiosis is that the blueprint of life,
the DNA, is provided in equal amounts to each of the daughter cells. The DNA in
all human cells except eggs and sperm is housed in 46 chromosomes. Eggs and
sperm consist of only half that number, 23 chromosomes per cell. Every person
inherits one set of chromosomes from their mother (in the egg) and the other
set of chromosomes from their father (in the sperm). It is imperative that when
mitosis and meiosis are complete, the appropriate number of chromosomes exists
in each cell. If there are extra or missing chromosomes, the cells usually do
not live. To ensure that there are enough chromosomes to be divided evenly
between the two daughter cells during mitosis, an essential step precedes cell
division that allows the chromosomes to be copied, a step called DNA
replication. In mitosis, (1) the DNA is replicated, (2) the chromosomes are
separated into two equal sets, and (3) the cell divides. These steps must occur
in this exact order or the resulting cells will have an incorrect number of
chromosomes and may not live.
A similar set of steps occurs in
meiosis: (1) the DNA is replicated, (2) the chromosomes are separated twice
into four equal sets, and (3) the cell divides into four cells. Each of these
daughter cells has to contain 23 chromosomes, half of the original 46. Again
these steps must occur in this exact order or the resulting cells will have an
incorrect number of chromosomes.
It has probably become obvious at
this point that mitosis and meiosis are not simple processes. In fact, there
are literally hundreds of genes that are needed to control the many steps that are
involved and to be sure that those steps occur in the correct order. Any
mistakes can be deadly. For example, it is necessary to the cell’s survival
that the DNA gets replicated and the chromosomes are separated equally so that
all daughter cells get the correct number of chromosomes. It is also necessary
to control the frequency at which cells divide; if cells divide more often than
they should, then cancerous tumors are often the result. The death rate due to
cancer in the United States dropped between 1991 and 1995 for the first time
since the 1930s, translating into the saving of 10,000-15,000 lives per year
(National Cancer Institute, 1999). Despite this decline in the cancer death
rate, however, the prognosis is grim. Over 550,000 Americans are expected to
die of cancer this year and just under half of all men and just over one-third
of all women will contract cancer sometime during their lives (American Cancer
Society, 2002). Cancer is currently the second leading cause of death, and, by
2004, cancer will be the number one cause of death in the United States
(American Cancer Society, 2002).
Although cancer is a very complex
disease, it has recently become clear that many of our very own genes that are
supposed to be working in our cells to control cell division (mitosis and
meiosis) are responsible for the cancer epidemic. These normal genes are
altered or mutated, possibly due to our exposure to such things as tobacco
smoke (which contains at least 50 known cancer-causing agents), UV light from the
sun (or from the tanning booth), certain preservatives in our foods, and a host
of other environmental assaults (American Cancer Society, 2002). When these
normal genes are mutated, they are no longer able to do their jobs effectively,
a problem that can lead to cancer.
It is very difficult to study such a
basic problem as cell division in humans, although some researchers do. It has
become increasingly common, however, to use model organisms, organisms that are
similar to humans in terms of how their genes work. One organism that is used
worldwide by thousands of researchers in the study of cell division is Saccharomyces cerevisiae. This organism
is a single-celled yeast, a fungus, and is the same one that is used to brew
beer and make wine and bread. Yeast grows quickly, producing a new generation
of cells in about two hours. (Human cells in culture take about 24 hours to
produce a new generation). It is easy to manipulate, cheap to maintain, and is
nonpathogenic. (Human cells die easily, require expensive equipment and media,
and special safety precautions must sometimes be taken to work with them).
Yeast have been studied by researchers for nearly 100 years, resulting in
innumerable experimental techniques that have been designed for use with this organism.
(There are many experiments that simply cannot be done with human cells because
the techniques have not been developed or they just don't work). Finally,
although yeast does not look anything like a human, it still grows and divides
by the processes of mitosis and meiosis and uses most of the same genes to
control those processes.
Two of the many genes that yeast and
higher organisms have in common are CDC7
and DBF4. The CDC7 gene was identified in yeast about 30 years ago (Hartwell,
1976) and was shown to be needed for the important DNA replication step in
mitosis. Since then, many research labs have discovered that this CDC7 gene actually switches DNA
replication "on" in mitosis in yeast. Yeast cells that have a
mutation in the CDC7 gene cannot even
start the DNA replication step and therefore become stuck or “arrested” in
mitosis as single cells that eventually die (Jackson, et al., 1993). Many research labs have become interested in
studying CDC7 and its role in
mitosis, especially since a human version of this gene was recently isolated
and found to be altered in tumor cells (Hess, et al., 1998), associating this gene with cancer formation.
The DBF4 gene was implicated as a partner for CDC7 in the process of DNA replication when Kitada and colleagues
obtained genetic evidence that the two gene’s protein products interact
(Kitada, et al., 1992). More recent
biochemical experiments have supported this hypothesis by showing directly that
the Cdc7 and Dbf4 proteins really do interact, and they do so only at the
initiation of DNA replication during mitotic growth (Oshiro, et al., 1999). It is clear from these
experiments that the Cdc7 and Dbf4 proteins work together to initiate DNA
replication during mitosis.
Although a fair amount is known about the roles of CDC7 and DBF4 in mitosis, research on meiosis has been far less intense.
Nearly 25 years ago, a paper was published that presented the results of
experiments designed to determine the role of CDC7 in meiosis in yeast (Schild and Byers, 1978). These scientists
concluded that although CDC7 was
needed for the chromosomes to replicate during mitosis, it was not needed for
the chromosomes to replicate during meiosis. How strange! Why would DNA
replication be controlled differently in mitosis and meiosis when the two types
of cell division are so similar otherwise? Although scientists who studied DNA
replication were confused by these results, the work done by Schild and Byers
seemed clear enough and few scientists tried to explain the paradox. Because of
this, papers about CDC7’s role in
meiosis have been scarce since 1978. Unbelievably, even less work has been done
on the role of the DBF4 gene in
meiosis. Kitada, et al. (1992) showed
that dbf4 mutants are unable to
complete meiosis and form spores, but that is the extent of published work on
the meiotic phenotype of dbf4.
My students and I have been working for the past few
years to begin to elucidate the roles of these two genes in meiosis. We have
made important advances and our work has been received with increasing interest
at national scientific meetings (e.g., Wheeler, et al., 2002). It is clear at these meetings that more and more
researchers are interested in the regulation of meiosis and the genes that are
involved in this regulation.
![]()
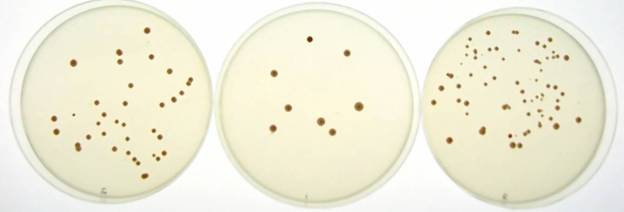
Figure
2. Colonies of yeast growing on specific media types in the lab. Each single
colony (for example, at the tip of the arrow) contains millions of individual
cells.
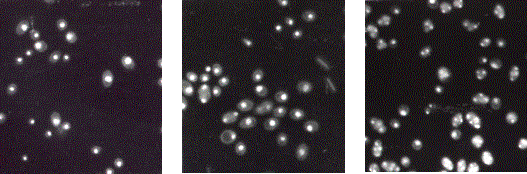
![]()
![]()
![]()
![]()
![]()
![]() Figure 3. Yeast cells
treated with DAPI, a dye that stains DNA and causes nuclei to fluoresce when
observed using a certain wavelength of light. These are wild-type sporulating
yeast cells sampled from different times during meiosis. The bright spots in
each cell are the DNA-containing nuclei. (A) All cells have only one bright spot
of DNA, indicating that the first division of the chromosomes has not yet
occurred in these cells. (B) One hour later, there are three cells out of those
in the photo that have two bright spots of DNA, indicating that these cells
have gone through the first of the two chromosome divisions. (C) Six hours
later, there is a mix of cells whose chromosomes have not divided (one spot of
DNA), divided once (two spots of DNA), or divided both times (four spots of
DNA). As expected for a wild-type strain, most have divided twice and therefore
have four spots of DNA. Photos taken from Galbraith (1995).
Figure 3. Yeast cells
treated with DAPI, a dye that stains DNA and causes nuclei to fluoresce when
observed using a certain wavelength of light. These are wild-type sporulating
yeast cells sampled from different times during meiosis. The bright spots in
each cell are the DNA-containing nuclei. (A) All cells have only one bright spot
of DNA, indicating that the first division of the chromosomes has not yet
occurred in these cells. (B) One hour later, there are three cells out of those
in the photo that have two bright spots of DNA, indicating that these cells
have gone through the first of the two chromosome divisions. (C) Six hours
later, there is a mix of cells whose chromosomes have not divided (one spot of
DNA), divided once (two spots of DNA), or divided both times (four spots of
DNA). As expected for a wild-type strain, most have divided twice and therefore
have four spots of DNA. Photos taken from Galbraith (1995).
References
·
American
Cancer Society (2002) [http://www.cancer.org/eprise/main/docroot/stt/stt_0]
·
Galbraith,
A. M. (1995). A genetic and molecular characterization of the REC104 gene and its involvement in
meiotic recombination in the yeast Saccharomyces
cerevisiae, UMI Dissertation Services.
·
Hartwell,
L. (1976). Sequential function of gene products relative to DNA synthesis in
the yeast cell cycle. Journal of Molecular Biology 104:803.
·
Hess,
G.F., R. F. Drong, K. L. Weiland, J. L. Sligthom, R. A. Sclafani, and R. E.
Hollingsworth, Jr. (1998). A human homolog of the yeast CDC7 gene is overexpressed in various tumors and transformed cell
lines. Gene 211:133.
·
Jackson,
A.. L., P. M. B. Pahl, K. Harrison, J. Rosamond, and R. A. Sclafani (1993).
Cell cycle regulation of the yeast Cdc7 protein kinase by association with the
Dbf4 protein. Molecular and Cellular Biology 13:2899.
·
Kitada,
K., L. H. Johnston, T. Sugino, and A. Sugino (1992). Temperature-sensitive cdc7 mutations of Sacchmaromyces cerevisiae are suppressed by the DBF4 gene, which is required for the
G1/S cell cycle transition. Genetics 131:21.
·
National
Cancer Institute (1999) [http://wwwosp.nci.nih.gov/bypass99/director/index.html]
·
Oshiro,
G., J. C. Owens, Y. Shellman, R. A. Sclafani, and J. Li (1999). Cell cycle
control of Cdc7p kinase activity through regulation of Dbf4p stability.
Molecular and Cellular Biology 19:4888.
·
Schild,
D. and B. Byers (1978). Meiotic effects of DNA-defective cell division cycle
mutations of Saccharomyces cerevisiae.
Chromosoma 70:109.
·
Wheeler,
J., S. Hanson, N. Krause, C. Zabel, R. Rohrer, and A. Galbraith (2002).
Determining the roles of the CDC7 and DBF4 genes in meiosis in Saccharomyces cerevisiae, poster
presentation, Yeast Genetics and Molecular Biology Meeting, Madison, WI, August
2002.